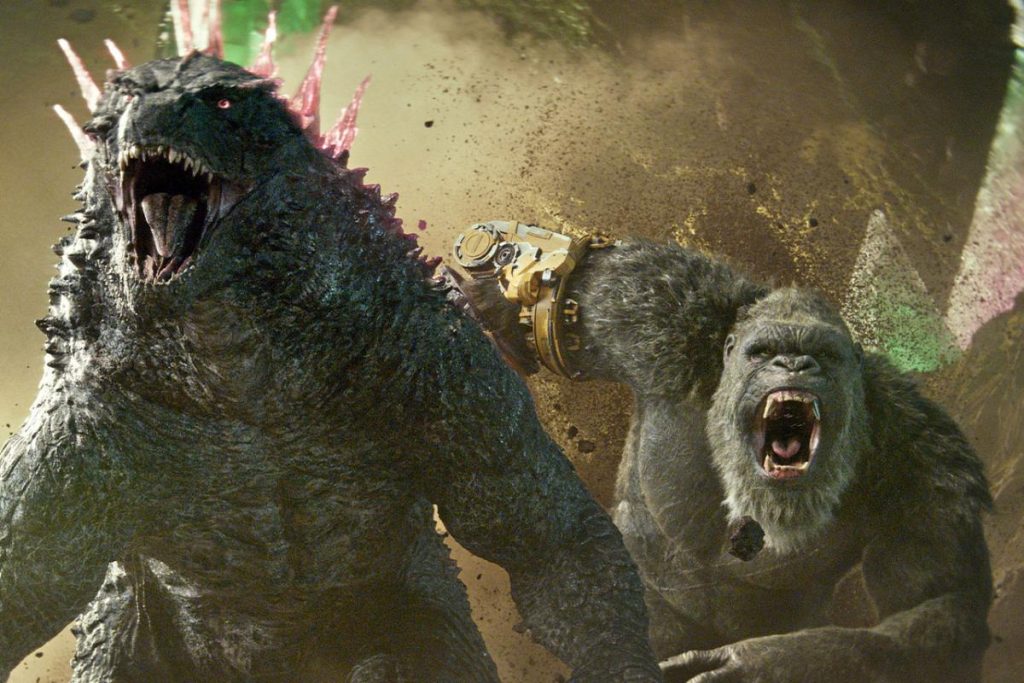Godzilla x Kong: The New Empire is the highly anticipated sequel to Godzilla vs. Kong, featuring Rebecca Hall and Brian Tyree Henry. The film is the 38th installment in the Godzilla franchise and the 13th in the King Kong franchise. In this new film, Kong and Godzilla must face a colossal undiscovered threat hidden within our world while exploring the mysterious origins of the two monsters.
Currently, the only way to watch Godzilla x Kong: The New Empire is by heading out to a movie theater starting on Friday, March 29. Viewers can find local showings on Fandango. Alternatively, the film will eventually be available to rent or purchase on digital platforms like Vudu, Apple, YouTube, and Amazon, as well as on streaming services like Max. Warner Bros. Pictures, the distributor of the movie, will release it on Max after 45 days from its theatrical release.
The streaming release date of Godzilla x Kong: The New Empire on Max is not officially confirmed yet. However, based on the release pattern of previous Warner Bros. movies, it is estimated that the film might not be available for streaming until late May 2024. Typically, Warner Bros. Discovery films head straight to Max after their theatrical release, but there is also a possibility that Netflix could obtain the streaming rights to the movie due to a deal between the two streaming platforms for certain Warner Bros. Discovery projects.
In conclusion, fans of Godzilla and King Kong can look forward to the epic showdown in Godzilla x Kong: The New Empire. While the movie will only be available in theaters initially, it will eventually make its way to digital platforms and streaming services for wider accessibility. Stay tuned for updates on the streaming release date, as Warner Bros. Pictures continues to navigate the changing landscape of movie distribution. Get ready to witness the clash of titans and uncover the hidden threats that await in the latest installment of this iconic monster franchise.